Plumbing is about picking the right material for the right job and knowing how much energy it takes to do anything with it. Every system design choice — copper or plastic, galvanised or stainless, insulated or not, heat pump or gas — comes back to materials science. Get the fundamentals straight and a whole block of exam questions become easy marks.
This is the last of the four Level 2 plumbing science deep-dives. For the others, see the pressure and force, thermal expansion, and heat transfer posts. For the wider revision strategy, pair it with the spaced repetition guide.
Density: the starting point
Density is mass per unit volume. How much stuff is packed into a given space.
The formula:
Density = Mass ÷ Volume
Units are kilograms per cubic metre (kg/m³) in SI, though you'll also see g/cm³ in some contexts.
The one density figure you need cold:
Water at 4°C has a density of 1,000 kg/m³.
One cubic metre of water weighs 1,000 kg. One litre of water weighs 1 kg. These two facts are worth committing to memory because they unlock a huge number of calculations.
Water's strange behaviour
Water is one of the few materials that doesn't simply get denser as it cools. Its density peaks at 4°C and then decreases as it cools further toward freezing. This is why ice floats on water, and why lakes freeze from the top down rather than the bottom up.
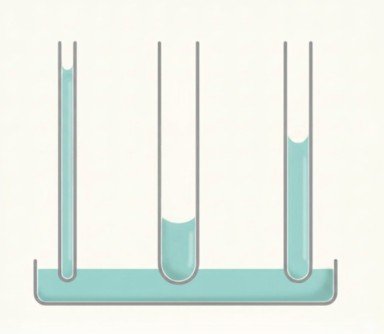
Three water expansion figures you need to know for the exam:
- Heated: water expands by about 4% when heated from 4°C to just below boiling (100°C)
- Frozen: water expands by about 10% when it freezes and turns to ice — which is why unlagged pipes in cold spaces burst
- Steam: water expands by about 1600 times when it turns to steam — the reason uncontrolled steam in a hot water or heating system is a genuine explosion risk
For exam purposes, the key fact is that water is at its maximum density at 4°C — and that it's unusual precisely because it expands in both directions from there.
Specific heat capacity
Specific heat capacity (SHC) is the amount of energy needed to raise 1 kilogram of a material by 1 degree Celsius. It measures how much heat a material can absorb per unit of temperature change.
The formula:
Q = m × c × ΔT
Where:
- Q = heat energy in joules (J) or kilojoules (kJ)
- m = mass in kilograms (kg)
- c = specific heat capacity in kJ/kg·°C
- ΔT = change in temperature in °C
The value you need to memorise:
Water's specific heat capacity ≈ 4.186 kJ/kg·°C (often rounded to 4.2)
Water has a very high specific heat capacity compared to most other materials. This is why it's used as a heat transfer medium in central heating — it holds a lot of energy per kilogram and releases it steadily as it cools.
Working the formula
A standard exam calculation: "How much energy is needed to heat 100 litres of water from 10°C to 60°C?"
- Mass: 100 litres × 1 kg/litre = 100 kg
- SHC of water: 4.186 kJ/kg·°C
- Temperature change: 60 − 10 = 50°C
- Q = 100 × 4.186 × 50 = 20,930 kJ (about 20.9 MJ)
That's a lot of energy — which is why heating a cylinder full of water takes time, and why hot water cylinder insulation saves so much fuel over a year.
Thermal conductivity
Thermal conductivity measures how readily a material conducts heat. In plumbing terms, it determines whether a material is useful for transferring heat (good conductor) or preventing heat transfer (poor conductor, i.e. insulator).
A working ranking for the materials that turn up in Level 2:
- Copper — very high thermal conductivity. Used where heat needs to move — heat exchangers, coils, cylinder immersion elements.
- Aluminium — also high, but corrodes too easily for most plumbing uses.
- Low carbon steel — moderately high. Good enough for radiators, where you want heat to pass through.
- Brass — similar to steel, often used for fittings and valve bodies.
- Plastics (PE, PEX, PVC) — low thermal conductivity. Good for cold water and drainage; less ideal for hot water distribution where heat retention matters.
- Insulation materials (foam, mineral wool) — very low conductivity. The whole point of insulation.
Thermal conductivity and specific heat capacity are different properties and regularly confused. Conductivity is how fast heat moves through a material; capacity is how much energy a material can store per kilogram per degree.
Ferrous and non-ferrous metals
Before the corrosion section — a term that comes up reliably in the exam.
Ferrous metals are metals that contain iron. Three things are true of ferrous metals:
- They contain iron
- They are magnetic
- They rust when exposed to air and water (this is called atmospheric corrosion)
The ferrous metals you'll meet at Level 2 are:
- Cast iron
- Low carbon steel (sometimes called mild steel)
- Malleable iron
A useful exception: stainless steel is NOT considered ferrous, even though it contains iron, because it has little magnetism and doesn't rust. The alloying elements in stainless steel change its properties enough that it sits outside the ferrous category for plumbing purposes.
Why does the distinction matter?
- Ferrous metals like cast iron and low carbon steel can only be used for heating circuits and drainage, because the rust they produce would contaminate a drinking water supply.
- Stainless steel and non-ferrous metals (copper, brass, bronze) can be used for potable water because they don't rust in the same way.
- In heating systems, a corrosion inhibitor is dosed into the system water specifically to protect the ferrous components (the steel radiators and any steel pipework).
Strength — three types
Different materials cope differently with the three types of mechanical force — tensile, compressive and shear. The diagrams and definitions of those forces are covered in the pressure and force post; what matters here is which materials handle each force type well.
- Tensile strength — resistance to being pulled apart. Steel, copper and most ductile metals are strong in tension. Cast iron and brittle ceramics are weak.
- Compressive strength — resistance to being crushed. Cast iron, concrete and stone are excellent in compression. Most ductile metals are good in compression too, but yield (deform) under heavy sustained load.
- Shear strength — resistance to being sliced. Steel and brass perform well; brittle materials like cast iron fail suddenly under shear, with little warning.
This is why fittings and components are made from specific materials. A brass compression fitting needs reasonable tensile and shear strength to handle pressurised flow; a cast iron drain cover needs compressive strength to take vehicle loads but isn't carrying tensile loads. Match the material to the load type the component will see in service.
Corrosion: the enemy of long-life plumbing
Metals in plumbing don't just sit there — they interact with water, oxygen, dissolved salts, and each other. The main forms of corrosion that come up at Level 2:
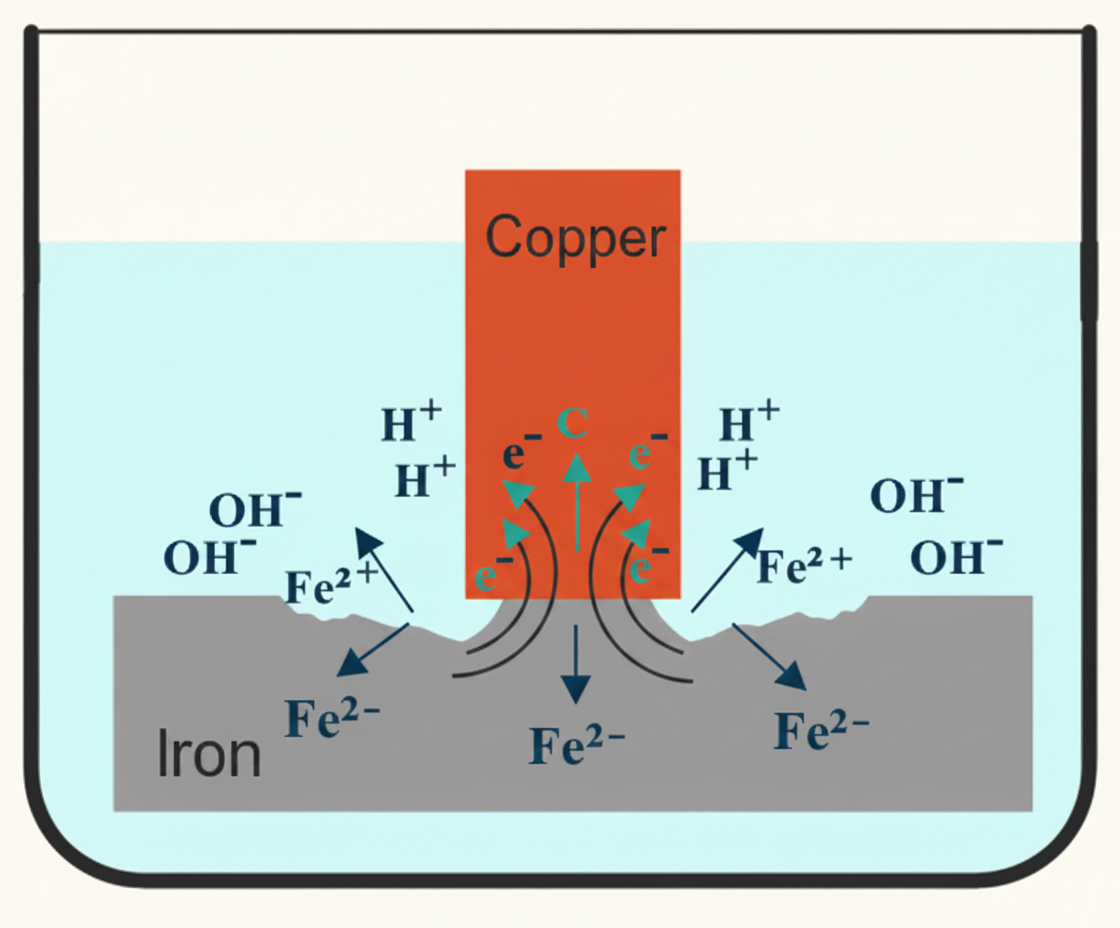
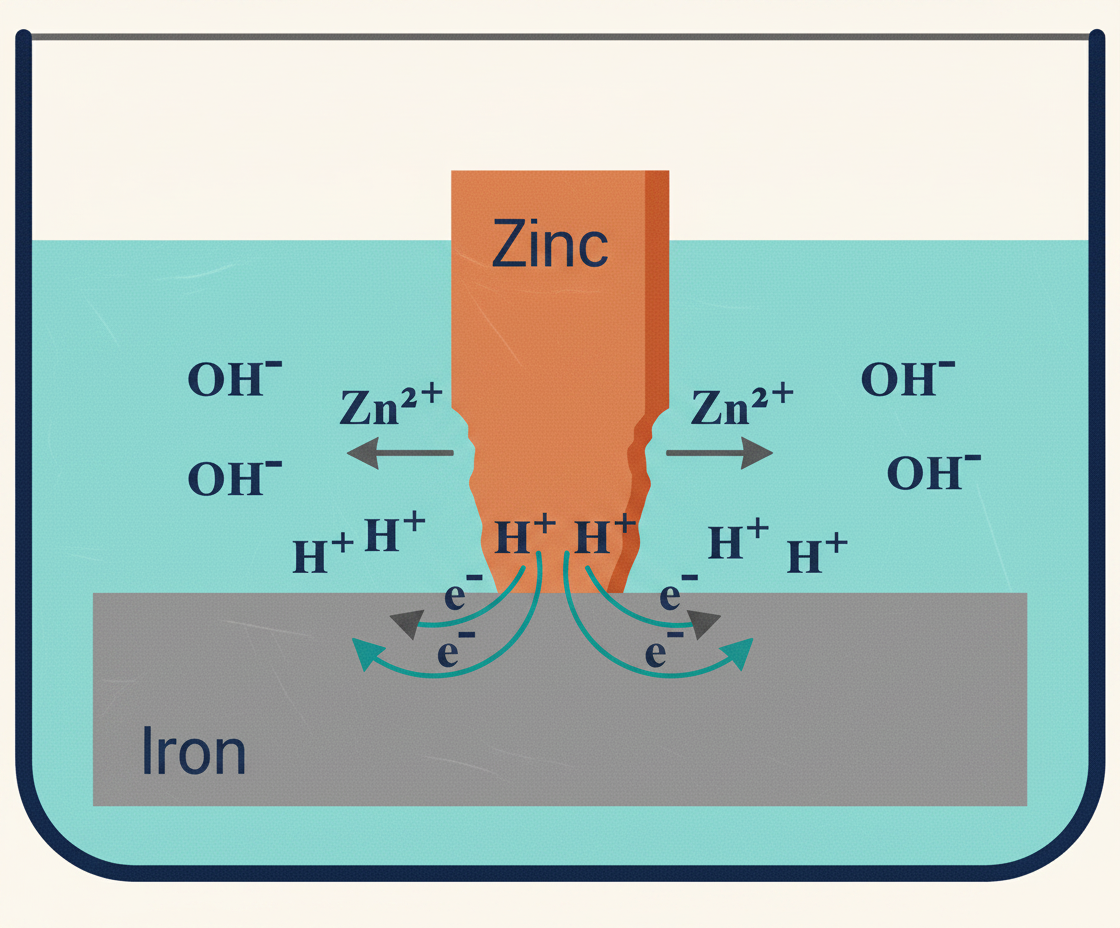
Electrolytic (galvanic) corrosion. When two different metals are in electrical contact via an electrolyte (water, damp air), one corrodes preferentially. Every metal sits somewhere on the electromotive series — a ranking from most anodic (most reactive, corrodes first) to most cathodic (most stable, corrodes last). The further apart two metals are on the series, the more aggressive the reaction between them.
The classic plumbing problem: copper joined directly to galvanised steel. Copper sits higher on the electromotive series than zinc, so the zinc coating on the galvanised steel is destroyed first. Once the zinc is gone, the steel underneath starts corroding too. Fix: use a fitting (such as a brass connector or dielectric coupling) that separates the two metals and breaks the electrical contact.
Pitting corrosion. Small, localised pits forming on the inside of copper pipe. Most common in hard water areas, but also caused by poor workmanship — particularly leaving excess flux on a soldered joint, which is acidic and attacks the copper over time. Fix: wipe excess flux off every joint after soldering.
Erosion corrosion. Found mainly in pumped hot water circuits where water velocity is too high, especially at bends, elbows and tees where turbulence is worst. The moving water gradually eats away at the pipe wall. Fix: design the system with appropriate pipe sizing so velocities stay within normal limits.
Blue water corrosion. When new copper pipework doesn't form its normal protective oxide layer — usually because water has stagnated in the system before use — copper leaches into the water, giving it a characteristic blue, cloudy appearance. Most common in new-build properties where pipes have sat unused. Fix: flush new systems thoroughly before first use.
Why material selection matters
Every material choice on a job is a trade-off:
- Copper is durable, strong, and conducts heat well — but it's expensive, corrodes in some soil conditions, and reacts galvanically with steel.
- Plastic (PE, PEX, PVC) is cheap, flexible, corrosion-proof, and non-conductive — but it expands more, can't be used with gas, and some types can't handle high temperatures.
- Low carbon steel is strong and cheap but corrodes readily — fine for radiators in a closed inhibited system, not for long-term water supply.
- Stainless steel is corrosion-resistant but expensive — used for commercial work and high-aggression environments.
Exam questions test whether you can match a material to a use and explain why.
Common exam traps
Trap 1: Confusing conductivity and capacity. Thermal conductivity is how fast heat moves through. Specific heat capacity is how much heat is needed to raise the temperature. Different questions, different answers.
Trap 2: Q = mcΔT unit confusion. The "c" value is typically given in kJ/kg·°C. If you plug in c = 4,186 (in J/kg·°C) you get an answer 1,000× too big.
Trap 3: Assuming all corrosion is the same. Electrolytic corrosion needs two different metals and an electrolyte. Pitting is often about workmanship (excess flux) or water hardness. Erosion corrosion is about water velocity. Blue water is about stagnation in new copper. Questions test the specific mechanism, not "corrosion" as a general category.
Quick revision summary
Before the mock test, seven things you need to be able to produce from memory:
- Density of water = 1,000 kg/m³ (peaks at 4°C)
- 1 litre of water = 1 kg
- Water expansion: 4% heated to near boiling, 10% when it freezes, 1600× when it turns to steam
- Specific heat capacity of water ≈ 4.186 kJ/kg·°C
- Q = m × c × ΔT — the heat energy formula
- Ferrous = contains iron, is magnetic, rusts. Cast iron, low carbon steel, malleable iron. Stainless steel is not ferrous.
- Copper + galvanised steel = electrolytic corrosion (the zinc corrodes first)
📝 10-Question Mock Test
Click an option to see whether you got it right. Explanations appear instantly — no submitting at the end.
The most important single density figure in the plumbing syllabus. Option A is a classic unit-confusion distractor (that's roughly the density of air, not water).
Water's peculiar property — density peaks at 4°C and decreases below that. Below 0°C it freezes and becomes ice, which is less dense than liquid water (which is why ice floats). A gift question if you've memorised the fact.
The value to know. Some workbooks round to 4.2; either figure would be accepted in most exams. The distractors all differ by factors of 10, which is typical of unit-confusion questions.
Q = 50 × 4.186 × 40 = 8,372 kJ. The distractors are all the same digits with different decimal places — which is exactly how this question is built to catch out students who get the arithmetic right but lose track of the magnitude. Sense-check: heating a big tank of water from 20 to 60°C needs a lot of energy — option A (83.72 kJ) is clearly too small, and the real answer has to be in the thousands of kilojoules.
Very high thermal conductivity is exactly why copper is used in heat exchangers and cylinder coils. The other three range from moderate (steel) to very low (plastic, insulation).
Two different metals (copper and the zinc on galvanised steel) in electrical contact with an electrolyte (water, damp) set up a galvanic cell. Copper sits higher than zinc on the electromotive series, so the more reactive zinc coating corrodes first — and once it's gone, the steel underneath starts corroding too. This is the classic textbook example of electrolytic corrosion in plumbing. Pitting (A), erosion (B), and blue water (D) are all real forms of corrosion but they're driven by different mechanisms — workmanship/water hardness, water velocity, and stagnation respectively.
Water is unusual in expanding when it freezes — by about 10%. This is why unlagged pipes in cold lofts burst in winter: the water inside turns to ice, expands by roughly a tenth of its volume, and the pipe can't contain it. Option B (4%) is the figure for water expanding when heated from 4°C to just below boiling — a classic distractor because both figures come up in the same topic.
The three-part definition of a ferrous metal: it contains iron, it's magnetic, and it rusts (atmospheric corrosion) when exposed to air and water. The three ferrous metals you'll meet at Level 2 are cast iron, low carbon steel, and malleable iron. Option D (stainless) is the trap: stainless steel contains iron too, but because it's alloyed with other elements that prevent rusting and give it almost no magnetism, it's not considered ferrous for plumbing purposes.
Water holds a lot of energy per kilogram per degree, so a central heating system can transport a lot of heat from the boiler to the radiators using a relatively small mass of water. Option D is wrong — water very much does corrode most metals in some form; that's why inhibitors exist.
Density × volume = mass. 1,000 kg/m³ × 1 m³ = 1,000 kg. One litre = 1 kg; there are 1,000 litres in a cubic metre; therefore 1 m³ = 1,000 kg. Three ways to the same answer.
How PlumbMate puts this into practice
Questions like these are exactly what PlumbMate drills you on — but with the spaced repetition engine doing the scheduling so you're not retesting yourself on the stuff you already know.
- Flashcards, not essays. One prompt, one answer — the format that research has consistently shown works best for active recall.
- Wrong answers are logged. Every question you get wrong goes into a dedicated collection that resurfaces more frequently in future sessions.
- The 3× rule. You need to get a question right three times before it clears — one lucky guess isn't enough.
- Explanations on every question. Like the ones above, but on every single question in the app.