Two topics that look unrelated but sit together in the cold water syllabus because both are about water behaviour in real-world conditions — how the chemistry of the water affects the pipework it flows through, and how the surrounding temperature affects whether the system keeps working. Level 2 expects fluency in both.
This is the fifth post in the Level 2 cold water sub-cluster. For the others, see the water sources and supply, direct vs indirect systems, cisterns, fluid categories and backflow, and commissioning posts.
What makes water hard or soft
Rainwater is naturally slightly acidic because it dissolves small amounts of carbon dioxide (CO₂) from the atmosphere while forming in clouds. That slight acidity is what soft water is — water that has dissolved CO₂ but hasn't picked up any rocks or salts. Soft water is chemically basic water with a slightly low pH.
When that rainwater flows over the ground, what it picks up determines whether it stays soft or becomes hard:
Soft water areas: water flows over rocks it can't dissolve — sandstone is the classic example — or sits on acidic terrain like moorland, where it dissolves more CO₂ from the atmosphere.
Hard water areas: water flows over rocks that dissolve into it — limestone and chalk are the two main culprits. The dissolved rock makes the water alkaline and gives it its "hardness".
Britain has both, varying with ground geology. Parts of London sit on chalk and have hard water; parts of Scotland and Wales sit on granite and have soft water.
Temporary vs permanent hard water
Hard water breaks down into two sub-types depending on what's dissolved in it:
Temporary hard water. Contains dissolved calcium carbonate. The defining feature: when heated above 65°C, the calcium carbonate comes out of solution and forms limescale — the chalky white deposit you see in kettles, shower heads, and hot water pipework.
Permanent hard water. Contains dissolved calcium sulphate. The defining feature: doesn't form limescale when heated. Permanent hard water is chemically similar to temporary hard water but the specific calcium compound doesn't precipitate out with heat.
The name "temporary" refers to how the hardness can be removed — by heating the water, which drops the calcium carbonate as limescale and leaves softened water behind. "Permanent" hard water stays hard even after heating.
How to tell if you're in a hard or soft water area
Two practical tests:

Litmus paper. Soft water is acidic, hard water is alkaline — litmus paper colour-codes the result. Acidic water turns red litmus paper stays red / blue turns red; alkaline water turns red litmus blue / blue stays blue.
Check the hot tap outlet. If you see limescale around the outlet of the hot tap, you're in a temporary hard water area (the limescale only forms above 65°C, which is hot water territory). No limescale = either a soft water area or a permanent hard water area — the test doesn't distinguish these, but it reliably identifies temporary hard water.
The problems each type causes
Soft water problems:

- Slightly acidic, so it's corrosive over time — can damage copper pipework and cause pinhole leaks. This is a slow effect (usually 30+ years) but real, and soft water areas see it disproportionately.
- Reacts well with soap — you don't notice this as a problem; it's actually an advantage.
Hard water problems (specifically temporary hard):
- Forms limescale above 65°C — blocks pipes, fills heat exchangers, chokes up kettles and shower heads
- Reduces heating efficiency — limescale on a heating element insulates the element from the water, so more energy is needed to reach the same temperature
- Can damage boilers catastrophically — particularly on combi boilers where the heat exchanger is small and limescale blockage can cause overheating
- Reacts poorly with soap — you need more soap and detergent in hard water areas, and soap scum forms rings around baths
Hard water benefits:
- Drinking hard water is believed to have health benefits — the dissolved minerals may be nutritional
- Protects copper pipework — the alkaline water doesn't corrode copper the way soft water does
Pinhole corrosion — specific to soft water
Soft, acidic water flowing through copper pipes over decades can gradually corrode the pipe wall from the inside. The corrosion starts as tiny pits and, over time, develops into pinhole leaks — small but persistent weeps from the pipe wall.
Pinhole corrosion is a classic soft-water problem — the water's acidity, rather than its hardness, is what drives it. Exam questions test this: "which water causes pin-holing in copper pipe?" The answer is acidic (soft) water.
Water softeners — base exchange
In hard water areas, a base exchange water softener can be installed to remove the hardness. How it works:
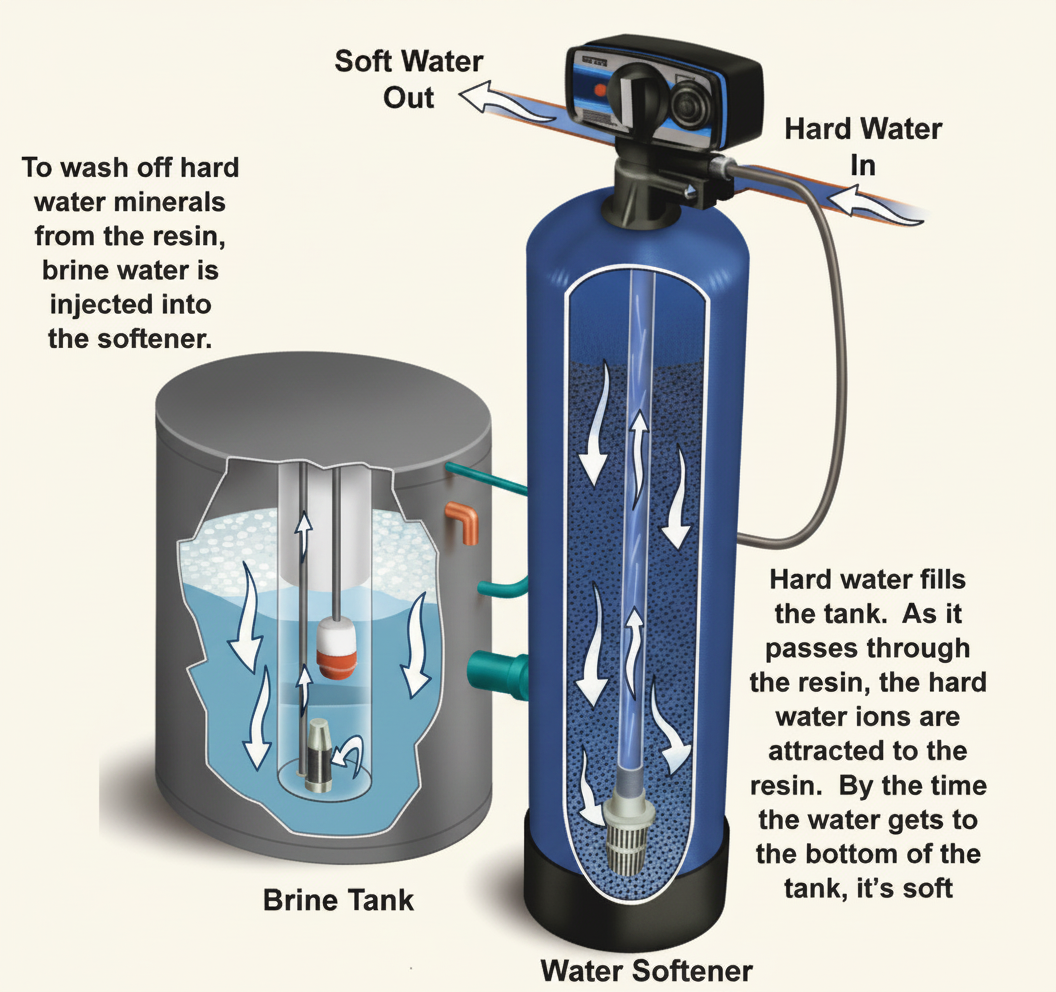
- The softener contains a resin bed charged with sodium ions
- Hard water flows through the resin
- The resin exchanges calcium ions (hardness) for sodium ions (softness) — the calcium stays behind in the resin, the sodium goes out with the water
- Eventually the resin becomes saturated with calcium and needs regenerating with salt (sodium chloride) — this is why water softeners need regular salt top-ups
The defining phrase: "exchanges the calcium ions for sodium ions" — if an exam question describes a water softener, this phrasing is the correct answer.
Other methods exist (electromagnetic, powerful acids, ultraviolet) but the workbook identifies base exchange as the standard method and these other options as incorrect on exam questions.
Output of a softener = Cat 2 water (aesthetic change from sodium content) — so a softener needs a single check valve on its output to prevent backflow.
The kitchen sink cold supply must not pass through a softener. Softened water is high in sodium, which isn't suitable for drinking, cooking, or anyone on a low-sodium diet. Modern installations install a bypass so the kitchen sink gets unsoftened mains water while everywhere else gets softened.
Frost protection — an overview
Water freezes at 0°C. When it freezes, it expands by about 10% (the fact that water expands rather than contracts on freezing is unusual among liquids). That expansion can split pipes, crack cisterns, and cause tens of thousands of pounds of damage in a single freeze/thaw cycle.
Frost protection strategies fall into three groups:
1. Burying pipes below the frost line. External supply pipes are buried minimum 750mm below ground level (covered in the supply to the property post). Below that depth, the ground temperature stays above freezing even in hard winters.
2. Insulation. Pipes and cisterns in unheated spaces get wrapped in pipe lagging (foam insulation). Insulation slows the rate at which heat is lost from the water, so even if the air temperature drops below freezing for a few hours, the water doesn't have time to freeze.
3. Active heating. Trace heating — an electric heating cable wrapped around the pipe, controlled by a thermostat. Switches on below a set temperature (typically around 5°C) and keeps the pipe warm enough to stop freezing. Used where insulation alone isn't enough, or on exterior pipework that can't be buried.
Frost protection in roof spaces
Roof spaces are the classic frost-risk location in UK properties — uninsulated attics can get very cold in winter. But they can also get very hot in summer (the roof above traps heat).
The rule: cold water supplies in roof spaces should be insulated for both frost AND heat protection. Not just frost. Not just heat. Both.
Practical consequences:
- Cisterns in the loft are insulated all around — the base, the sides, and the top — to prevent summer heat raising the water temperature above 25°C (bacterial growth threshold) and to prevent winter freezing
- Pipework to cisterns and hot water cylinders is lagged throughout its run in the loft
- The insulation under the cistern base is usually omitted so warm air from below can rise and help prevent freezing on the coldest nights
This last point is specific to cisterns: in the coldest conditions, a small amount of heat rising from the rooms below (through the unisulated ceiling directly under the cistern) can prevent the cistern freezing. The pipework still needs lagging; only the space directly beneath the cistern is left unisulated.
External pipework and taps
External pipework is particularly exposed to frost. Three protective measures:

- Outside taps — fit a service valve inside the property so the outside tap can be isolated and the external section drained for winter. Service valves on outside taps are good practice rather than regulatory but strongly recommended.
- Hose union bib taps — drain the hose from the tap after use in cold weather
- External supply pipes that enter the property above ground level — insulate the exposed section, not just the buried section
Common exam traps
Trap 1: What causes pinhole corrosion in copper pipework? Acidic (soft) water. Not hard water, not water with calcium, not alkaline water. Acidic = soft = pinhole corrosion.
Trap 2: Limescale forms above what temperature? 65°C. Below that, calcium carbonate stays dissolved; above it, limescale precipitates out.
Trap 3: Base exchange water softeners. They exchange calcium ions for sodium ions. Not "remove using acids", not "stabilise using electromagnetic fields", not "destroy with UV light". Exchange. Sodium for calcium.
Trap 4: Temporary vs permanent hard water. Temporary = calcium carbonate = forms limescale when heated. Permanent = calcium sulphate = doesn't form limescale. Temporary is the one that causes the problems.
Trap 5: Roof space insulation. Both frost AND heat protection. Not just frost.
Trap 6: Covering vs ventilating. The method of preventing pipes freezing is covering with insulation. Not ventilation, not pressure adjustment — insulation.
Trap 7: Trace heating threshold. Typically around 5°C — same as a frost thermostat. Covered in more detail in the heating controls post.
Quick revision summary
Before the mock test, seven things you need to be able to produce from memory:
- Soft water = acidic, forms on sandstone/moorland, causes pinhole corrosion in copper over decades
- Hard water = alkaline, forms on limestone/chalk, reacts poorly with soap
- Temporary hard = calcium carbonate, forms limescale above 65°C
- Permanent hard = calcium sulphate, doesn't form limescale
- Base exchange water softener = exchanges calcium ions for sodium ions
- Frost protection: bury pipes below 750mm (below frost line); insulate exposed pipes; trace heating for exterior pipework
- Roof spaces: frost AND heat protection on cold water supplies
📝 10-Question Mock Test
Click an option to see whether you got it right. Explanations appear instantly — no submitting at the end.
Soft, acidic water gradually corrodes copper pipework from the inside over decades, eventually creating pinholes. Options A, B and C describe hard water or chlorinated water, which don't cause this problem — in fact, alkaline (hard) water is protective against copper corrosion.
Calcium carbonate dissolved in temporary hard water precipitates out above 65°C, forming limescale on heating elements and inside pipes. Option A is a soft water problem (pinhole corrosion); C and D are unrelated.
The specific threshold above which calcium carbonate drops out of solution. Below 65°C the calcium stays dissolved; above it, limescale starts forming. This is why hot water cylinders in hard water areas are typically set at 60°C — just below the limescale threshold.
The defining principle of base exchange. The resin gives up sodium and takes calcium, swapping the two ions. Option A describes an electromagnetic conditioner (questionable effectiveness). Options B and D describe methods that aren't used in domestic softening.
Insulation is the standard primary protection — slows heat loss so water stays liquid even when air temperature drops. Trace heating (A) is a backup for situations where insulation alone isn't enough. Ventilation (C) would actually increase the freezing risk. Pressure (D) doesn't affect the freezing point in any useful way.
Loft spaces swing between freezing winters and overheating summers. Insulation protects in both directions — preventing freezing in winter and preventing summer heat pushing stored water above 25°C (the bacterial growth threshold from the cisterns post). Options A and B address only one risk.
Sandstone doesn't dissolve in water, so water flowing over it stays soft (acidic). Limestone and chalk (A, B) dissolve and make water hard. Mineral springs (D) typically contain lots of dissolved minerals and produce hard water.
Regular table salt (sodium chloride) is used to regenerate the resin in a base exchange softener. The sodium from the salt recharges the resin bed; the chloride and displaced calcium go to waste. Options A, C and D aren't used for regeneration.
Limescale only forms from temporary hard water (calcium carbonate) when heated. If there's limescale on the hot tap, the water contains calcium carbonate and is temporary hard. Soft water (A) would leave no limescale; permanent hard water (B) doesn't form limescale even when heated; softened water (D) has had the calcium removed.
Softened water is high in sodium, which isn't suitable for drinking, cooking, or anyone on a low-sodium diet. The kitchen sink cold supply should bypass the softener and get unsoftened mains water directly. Baths, showers and WCs all benefit from softened water (no limescale, better with soap).
How PlumbMate puts this into practice
Hard and soft water content combines some principles (why the chemistry works the way it does) with some fact-learning (which salt, which softener principle, which temperature). Spaced repetition handles both together.
- Flashcards, not essays. One prompt, one answer — the format that research has consistently shown works best for active recall.
- Wrong answers are logged. Every question you get wrong goes into a dedicated collection that resurfaces more frequently in future sessions.
- The 3× rule. You need to get a question right three times before it clears — one lucky guess isn't enough.
- Explanations on every question. Like the ones above, but on every single question in the app.